
Hi! My name is Daniela Ungureanu and I am a Co-PI of Jussi Taipale’s group at the University of Helsinki. I joined this position in January 2020, coming from Tampere University where I was an Academy Research Fellow and PI of the Cancer Signaling Group. My research background is in molecular and cellular biology, with a specific interest in protein phosphorylation, (pseudo)kinases and cell signaling dysregulation in diseases such as cancer. Along this research line, I am also actively working in functional precision oncology approaches to develop ex vivo patient-derived models for preclinical research.
“The importance of who you Kinome”
What is the Kinome?
The human kinome comprises of over 520 proteins, namely kinases, the enzymes that carry-out protein phosphorylation, one of the most exploited forms of post-translational modifications in the eukaryotic system. Interestingly, around 10-15% of kinases have one or more intrinsic mutations in their otherwise very conserved motifs required for proper catalytic activity, and consequently, were termed pseudokinases. Due to their mutated status, pseudokinases were basically outcast from our research interest compared to their active counterparts, until over a decade ago when several research breakthroughs highlighted their biological and therapeutic importance.
“Mechanisms, development, targets and interventions”
What are psudeo-kinases and what is there to understand from them?
HER3 for instance, is a receptor with high relevance in several cancers, but few of us know that HER3 is actually a pseudokinase. My interest in pseudokinases and their role in diseases started over 15 years ago, when I worked as a postdoctoral researcher on JAK pseudokinase domains that are hotspots for mutations linked to common myeloproliferative diseases such a polycythemia vera. I then moved on to work on ROR1 and ROR2 receptor pseudokinases, which are Wnt-ligand binding proteins involved in the onset of many cancers. My focus is to understand the molecular mechanisms associated with pseudokinase-mediated pathogenic signaling and to develop pharmacological approaches to target these proteins for therapeutic interventions. Within this goal, one needs to develop appropriate tools to test these molecules, and unlike protein kinases where we can measure phosphorylation activity more directly, monitoring pseudokinases and their activation mechanisms requires different approaches, as these proteins are (mostly) catalytically impaired. More challenges equal more innovation!
“Tailor-made treatments”
How does this research affect cancer patients?
Precision Oncology means that cancer treatment is specifically designed and targeted to a patient’s unique type of cancer. Unlike the conventional way of ”one size fits all” where we apply chemotherapy-based treatment that kills fast-dividing cells such as cancer cells, among many other healthy cells, precision oncology is the science of using patients’ own cancer cells to perform molecular profiling and in turn tailor the treatment to fit his/her so-called molecular signature.
“We’re gonna need the Omics..”
How would this need to be executed?
To do this, we need to have tumor cells undergoing molecular profiling so that we can fingerprint the pathogenic aberrations. Many patients undergo fine-needle biopsies for histologic diagnosis, thus the available tumor material for molecular profiling is not very abundant. Therefore, it is important for us to be able to expand the cancer cells ex vivo and to obtain sufficient material in due time for molecular profiling. This should optimally include several ”omics”, apart from NGS, like proteomics and functional omics such as HTP drug sensitivity and resistance testing. I have been using the HTB Unit at FIMM for several years now, to do large scale drug testing with patient-derived cancer cells in order to understand the pharmacological profiling of ex vivo patient-derived cancer cells grown in different conditions.
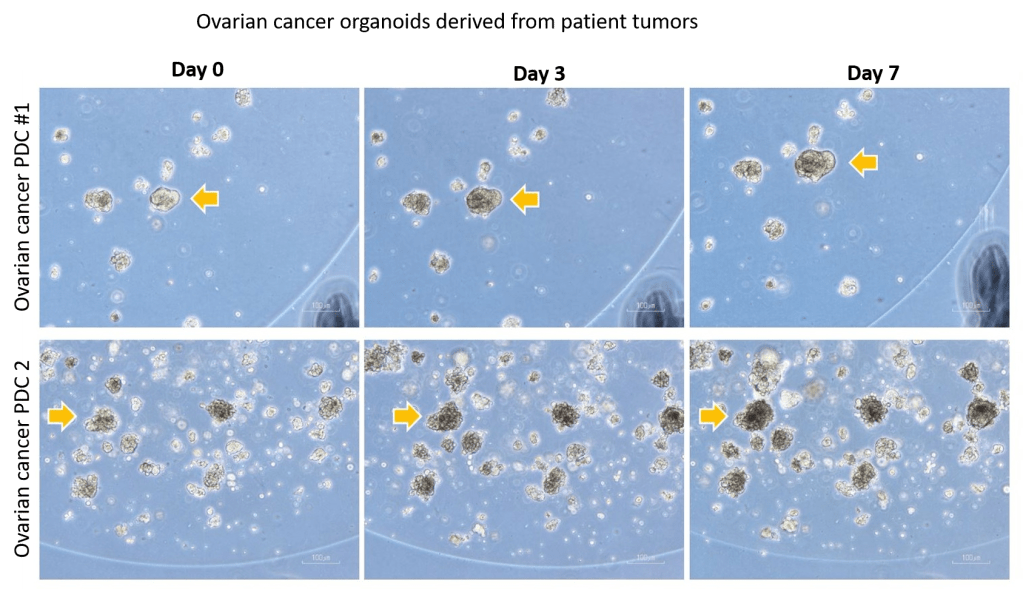
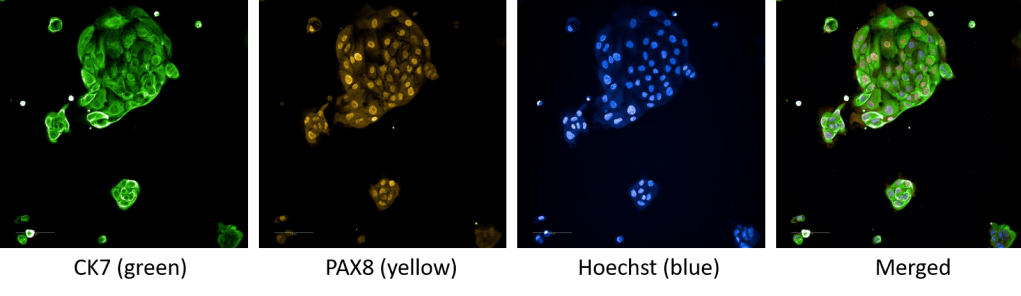
For instance, we have developed protocols to grow primary tumor cells in 2D, 3D (or spheroids) and lately as organoids. It is important to develop ex vivo culturing methods that mimic as closely as possible the tumor microenvironment, so that we can understand accurately how cancer cells grow and how we can kill it.
What is the future for this research?
Currently, we are growing long-terms patient-derived organoids from ovarian cancer patients undergoing treatment at HUS to perform HTP drug testing so that we can find drug vulnerabilities that could be clinically useful.
I believe the future will be driven more towards precision oncology, especially with our fast progress in lab technologies that allow us to isolate, grow and manipulate patient-derived cancer cells in lab.
Want to learn more about the Finnish Center of Excellence in Tumor Genetics?
Find the latest tweets, videos and blog posts for a peek into the everyday life in cancer research
by following us on Instagram (@tumorgenetics) and Twitter (@CoEinTG)
You can also subscribe to our YouTube channel!